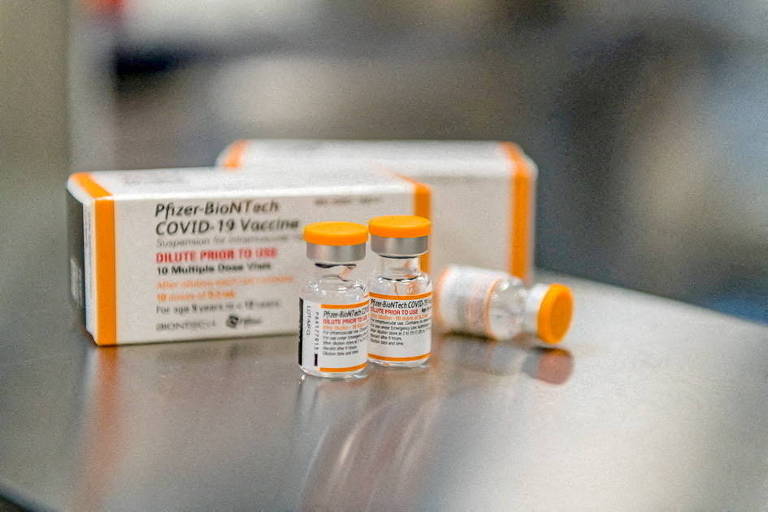
vaccine Pfizer And BioNTech is 73.2% effective in prevention COVID-19 Among children aged 6 months to 4 years, new data from companies showed this Tuesday (23), two months after the start of immunization implementation in United State for that age group.
The Pfizer-BioNTech vaccine was licensed for children under five in June in the United States, based on data showing that the vaccine elicited an immune response similar to that of older age groups.
An initial analysis based on ten asymptomatic cases of Covid-19 in the study suggested the vaccine was 80.3% effective. But experts cautioned that the data was preliminary due to the small number of symptomatic cases.
Updated data released Tuesday showed 13 children contracted Covid-19 at least seven days after receiving a third dose of the Pfizer-BioNTech vaccine, compared to 21 cases among those who received a placebo.
Most of the cases were due to Variant of Omicron Bachelor 2which was prevalent in March and April, when the study was conducted.
Pfizer and BioNTech also said they are preparing a US license application for a so-called bivalent vaccine targeting omicron’s BA.4/BA.5 sub variants for children under 12 years old.
The companies on Monday applied for a US license to the bivalent vaccine as a booster for people 12 years of age or older.

“Music fanatic. Professional problem solver. Reader. Award-winning tv ninja.”

/https://i.s3.glbimg.com/v1/AUTH_bc8228b6673f488aa253bbcb03c80ec5/internal_photos/bs/2024/V/A/331F8qSPGcp8g41HUPAw/gettyimages-2150216534.jpg)




More Stories
The “Gate of Hell” in Turkmenistan was created by human error; The gap reaches more than 400 degrees – the world
Jalisi mocks the TikTok siege in the United States: “Defenders of freedom?”
It's #Fact: A three-meter-long crocodile was found between the tires of military aircraft and was captured in the United States of America; Video | Real or fake